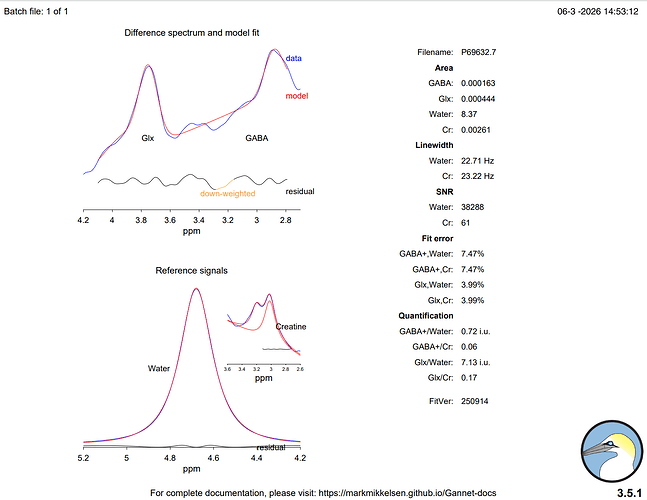
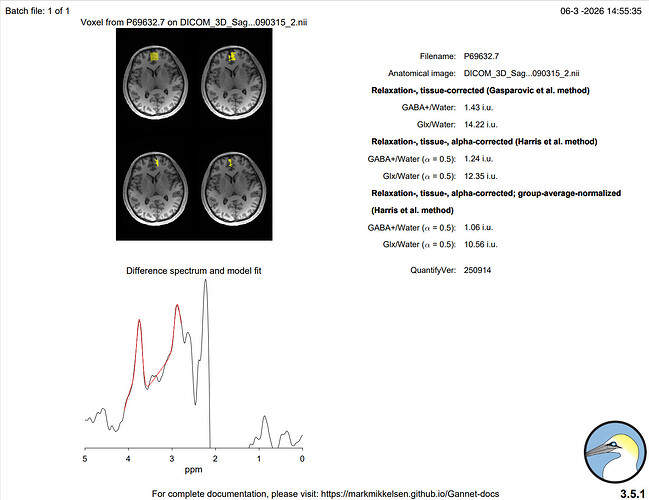
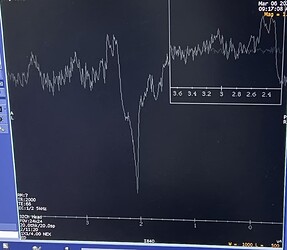
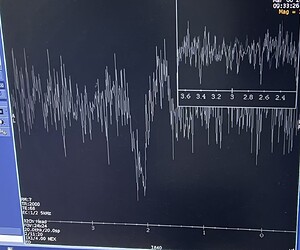
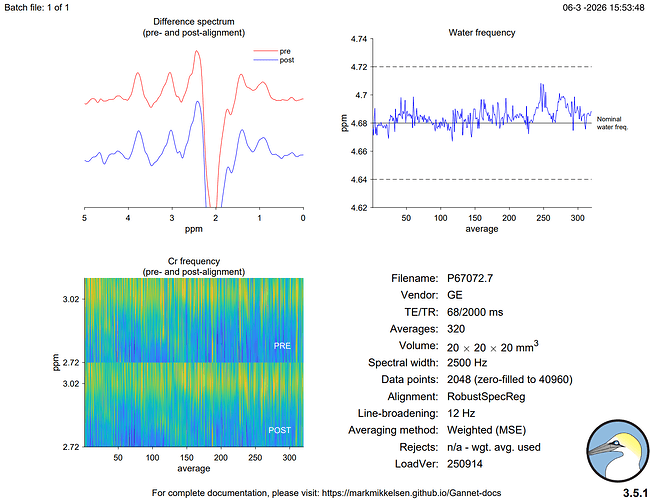
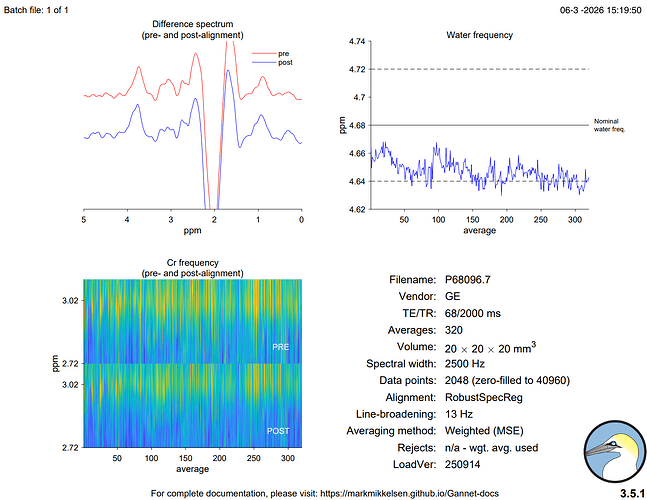
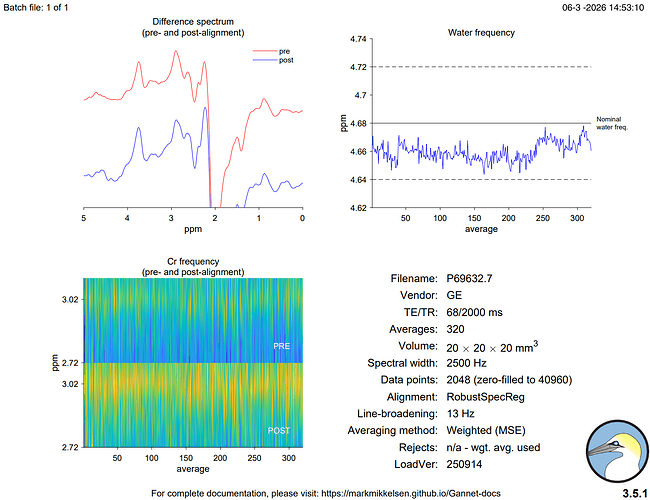
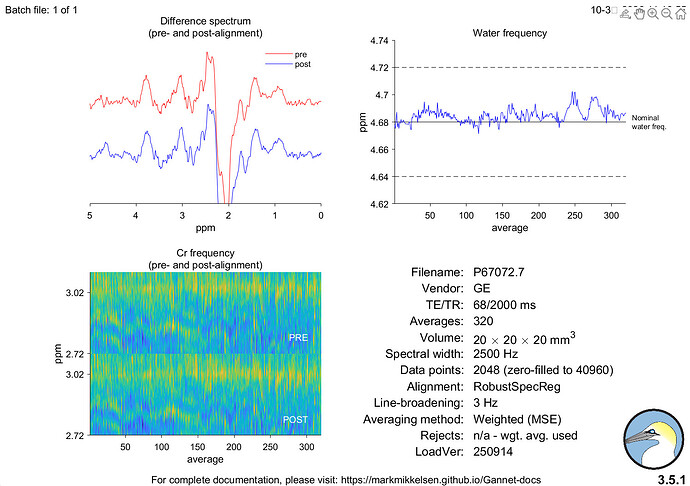
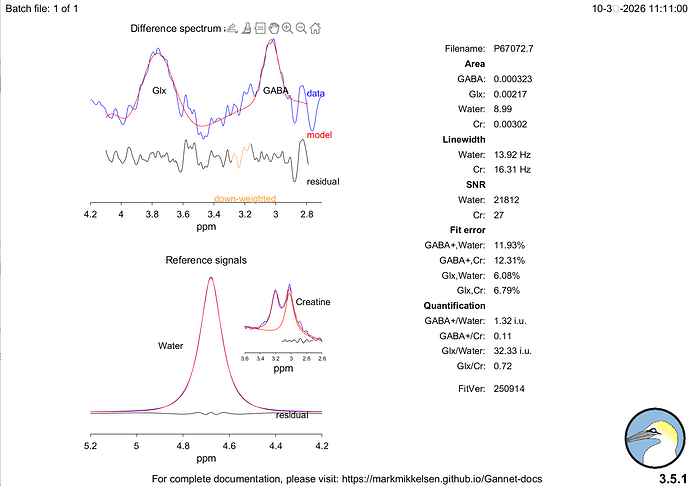
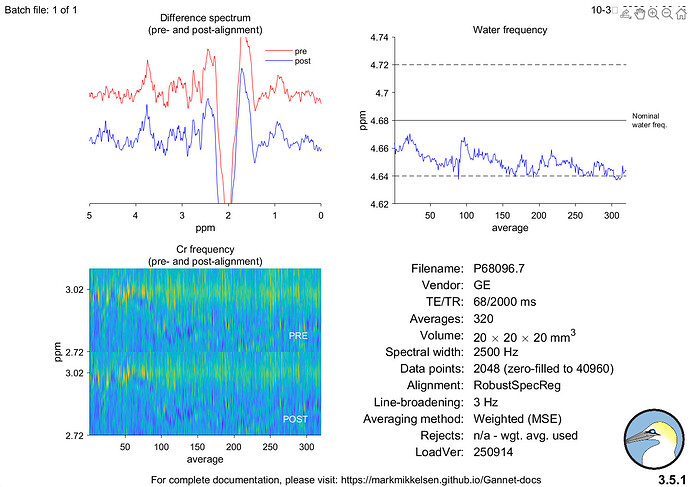
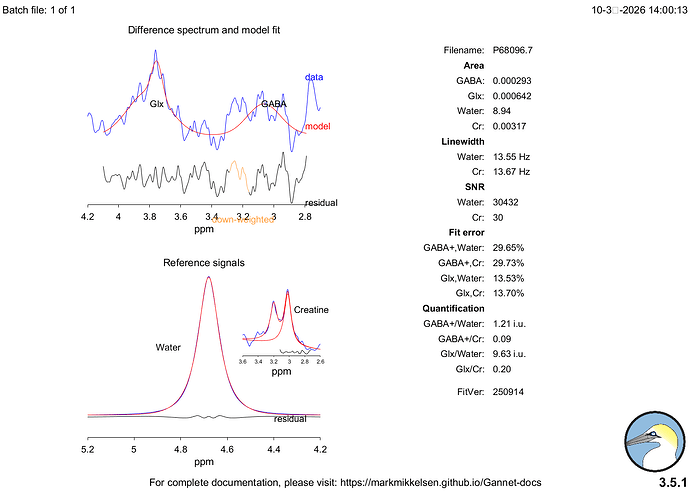
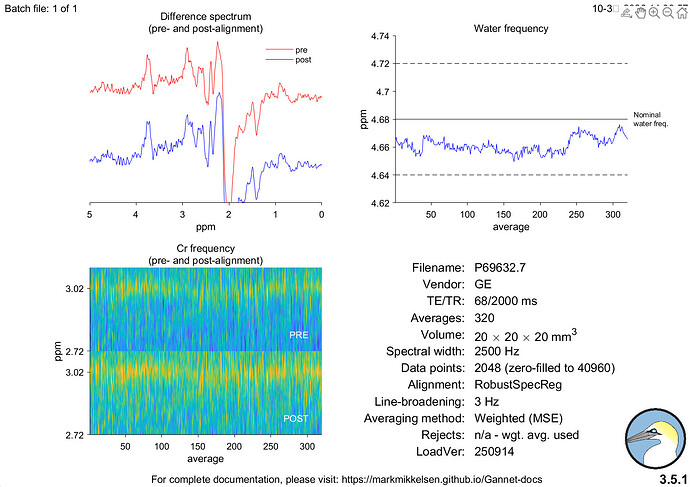
Hi everyone,
I’m planning an MRS study combined with noninvasive neuromodulation (e.g., tACS), and I’d really appreciate advice from anyone with experience or relevant references.
I have two main questions:
1. Is it meaningful to acquire MRS during stimulation (online MRS)?
For neurotransmitters/metabolites (e.g., GABA, Glu/Glx), do changes typically appear more reliably as offline/aftereffects rather than during the stimulation period? In other words, is “mid-stimulation MRS” expected to capture physiologically interpretable changes, or is it often dominated by non-specific factors (state changes, attention/arousal, etc.) and low sensitivity?
And although I’ve read some articles found the online effect by MRS(mostly tDCS), I still wonder whether neuromodulation can have effect on neurotransmitters in that short time and while MRS can capture that change.
2. How much does stimulation hardware/current affect MRS data quality (artifacts)?
For example, with high-frequency currents or typical tACS setups, do you see substantial MRS artifacts (RF interference, line noise, spurious peaks, frequency/phase instability, B0 drift, degraded linewidth/SNR, etc.)?
I’m aware that concurrent stimulation can strongly affect BOLD/fMRI signals—does something similarly problematic happen for MRS, or is it generally manageable with proper setup (cable routing, filtering, synchronization, pausing stimulation during readout, etc.)?
If you have practical recommendations (sequence choices, QC metrics to watch, whether to interleave ON/OFF blocks, filtering/shielding strategies, vendors/devices that work better, or “don’t do it” warnings), I’d be very grateful.
Thanks in advance!